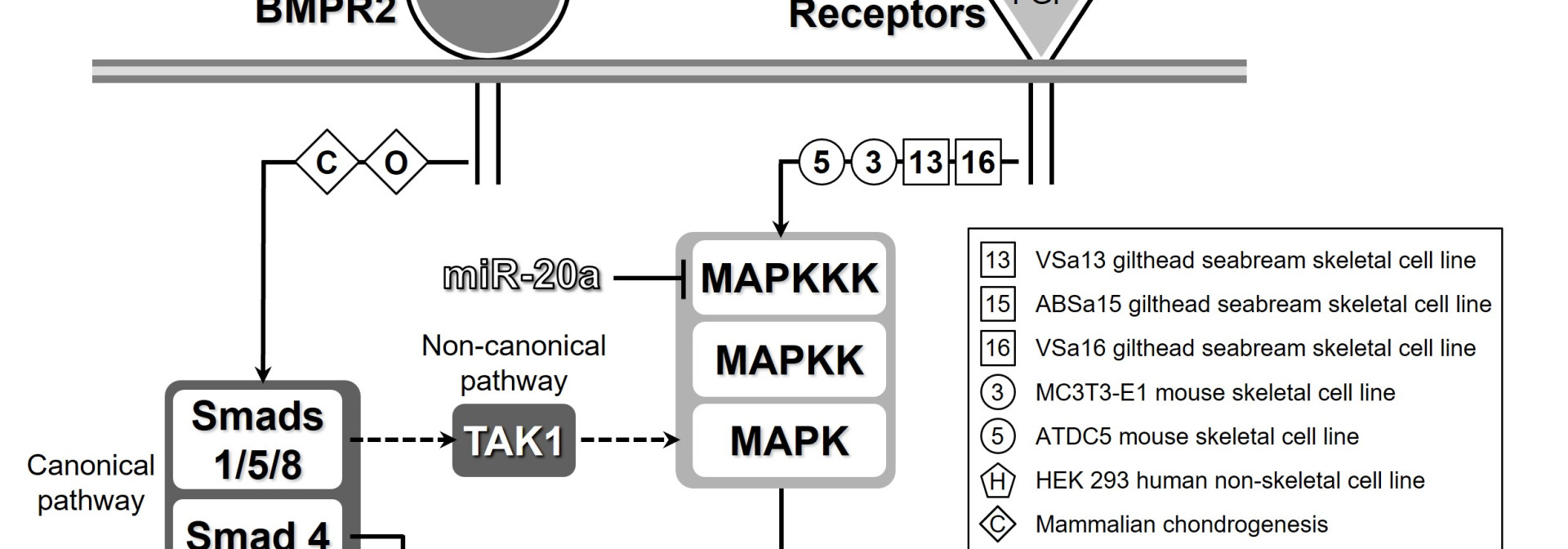
Our understanding of the mechanisms driving vertebrate skeletogenesis has been largely hampered by the complexity of the physiological processes and the numerous cellular and molecular factors involved. In particular, the transcriptional and post‑transcriptional regulation of skeletal gene expression by microRNAs (miRNAs) is likely to be of primary importance although still largely unknown. The main objective of this project is to identify microRNAs involved in skeletogenesis and characterize their target genes. To achieve this objective, we propose to use the zebrafish as a model for vertebrate skeletogenesis. An in vitro approach, based on recently developed bone-derived cell lines, and an in vivo approach, based on zebrafish exhibiting skeletal deformities, will be pursued in parallel. microRNAs differentially expressed during in vitro mineralization or in deformed fish, and therefore likely to play a role during tissue mineralization and skeletogenesis, will be identified using a microarray containing zebrafish miRNAs. Levels and sites of expression will be determined using an adapted qPCR method based on primer‑extension combined with LNA primers and an adapted in situ hybridization (ISH) protocol based on LNA probes labelled with fluorescein. Transcriptional regulation of miRNAs will also be investigated using luciferase reporter vectors containing the promoter region(s) of miRNAs identified through the microarray. Skeletogenesis- and mineralization-related function of selected miRNA will be evaluated using transgenic fish or cells where miRNAs gene expression has been altered. Phenotype will be determined using histological staining, immuno-histochemistry and bone mineral density measurements. Finally, miRNA target gene(s) will be identified by hybridizing available zebrafish DNA microarray with RNA from transgenic fish and cells, where expression of selected miRNAs has been modified. Data collected within the scope of this project should provide new insights into the mechanisms regulating vertebrate skeletogenesis.